Researchers have developed a 3D bioprinter for cancer research that can be folded into a carry-on pack, transported, and easily reassembled for printing inside a biosafety cabinet. The technology can perform rapid and reproducible manufacture of complex tumouroids – 3D cell cultures which resemble tumour tissues – to test potential immunotherapy treatments.
BioArm has the potential to screen a wide range of tumour therapies and could play a crucial role in the future development of cancer drug testing approaches.
Research Associate Corrado Mazzaglia
‘BioArm’ is a low-cost extrusion-based bioprinter to rival existing open-source bioprinters that cost less than £1,000. Small and lightweight (around 5kg), BioArm is based on a custom-built printhead and a hackable robotic arm. It has an average print time of approximately 90 seconds per tumouroid. During the robotic arm movement, bio-ink – in this case a cell-laden hydrogel containing samples that simulate mice tumours – is extruded from the printhead to form the desired 3D soft material constructs on a petri dish.
A cross-disciplinary research team from the University of Cambridge and King’s College London1 studied the interaction between immune cells and the reconstructed tumouroids with cancer-associated fibroblasts (CAFs), and then subsequently simulated immunotherapy treatments. Details of their approach are reported in the journal Biofabrication.
Immunotherapy is a form of cancer treatment that uses the body’s immune system to identify and fight cancer cells. When it comes to in vitro cancer modelling for therapeutic testing, increasing efforts are being made to mimic the tumour microenvironment, which also include fundamental stromal components – an important part of the body’s immune response. While animal models are frequently used to assess new treatment methods in cancer research, modelling a tumour in vitro can help reduce animal model use in the future. Due to its precision and reproducibility, 3D bioprinting has already been shown as a valuable tool to help construct in vitro cancer models of this nature, but most existing bioprinters are difficult to move around or reassemble. According to the researchers, BioArm, in comparison, has been designed to be portable for easy use of storage and transportation, and can be assembled and disassembled within 15 minutes.
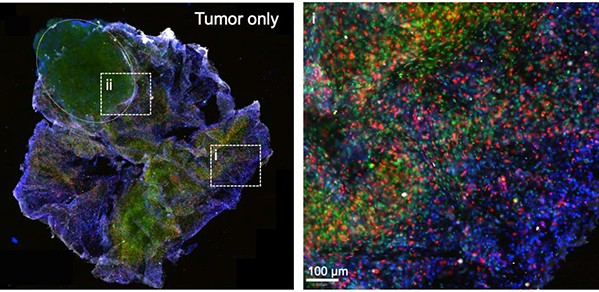
To assess if BioArm’s 3D printed compartmental tumouroids could be applied to test potential therapies as part of drug discovery pipelines, the researchers used live cell imaging and image analysis to study immune cell migration. This was conducted in 3D collagen gels in the presence of the tumouroid. High resolution imagery successfully captured the immune cells infiltrating the 3D printed tumouroids, with the immune cells stimulated with immunotherapy moving noticeably faster.
“BioArm, with its portability and ease of use, has the potential to make 3D bioprinting accessible to a wider research community,” said co-first author Yaqi Sheng, PhD student in the Biointerface Research Group at Cambridge. “This bioprinter has broken new ground; it can readily adapt to different working environments, while preserving relatively high accuracy. Its adaptability, customisation flexibility and biological relevance has not yet been shown by existing low-cost and open-source bioprinters.”
Corrado Mazzaglia, first author of the paper and Research Associate in the Biointerface Research Group, added: “Bioprinted cancer models mimic the 3D heterogeneity of real tumours. BioArm has the potential to screen a wide range of tumour therapies and could play a crucial role in the future development of cancer drug testing approaches.”
Reference:
Corrado Mazzaglia., Yaqi Sheng., Leonor Nunes Rodrigues., Iek Man Lei., Jacqueline D. Shields., Yan Yan Shery Huang. ‘Deployable extrusion bioprinting of compartmental tumoroids with cancer associated fibroblasts for immune cell interactions’. Biofabrication (2023). DOI: 10. 10.1088/1758-5090/acb1db
- - -
1 From the MRC Cancer Unit, The Nanoscience Centre, and the Department of Engineering (University of Cambridge) and the Comprehensive Cancer Centre (King's College London).

